What's the Spodumene Lithium Extraction Process?
 Shirley
Shirley
 Jul 13, 2023
Jul 13, 2023
 5530
5530
If you want to know more details about equipment, solutions, etc, please click the button below for free consultation, or leave your requirements!

The demand for lithium has been on the rise, fueled by the increasing use of lithium-ion batteries in electric vehicles, portable electronic devices, and renewable energy storage systems. As a result, the lithium industry is continuously seeking more efficient, cost-effective, and environmentally friendly methods for extracting lithium from various sources. One such source is spodumene, a lithium-bearing mineral found in pegmatite rocks. This article will explore the spodumene lithium extraction process, focusing on the innovations and challenges associated with this method.
01Spodumene Lithium Composition
BackSpodumene is a pyroxene mineral composed primarily of lithium, aluminum, and silica (LiAlSi2O6). It is one of the most abundant and economically viable lithium-bearing minerals, accounting for approximately 70% of the world's lithium reserves. Spodumene is found in pegmatite formations, which are coarse-grained igneous rocks with large, interlocking crystals. The mineral's high lithium content and widespread distribution make it an attractive target for lithium extraction.
02Traditional Spodumene Lithium Extraction Process
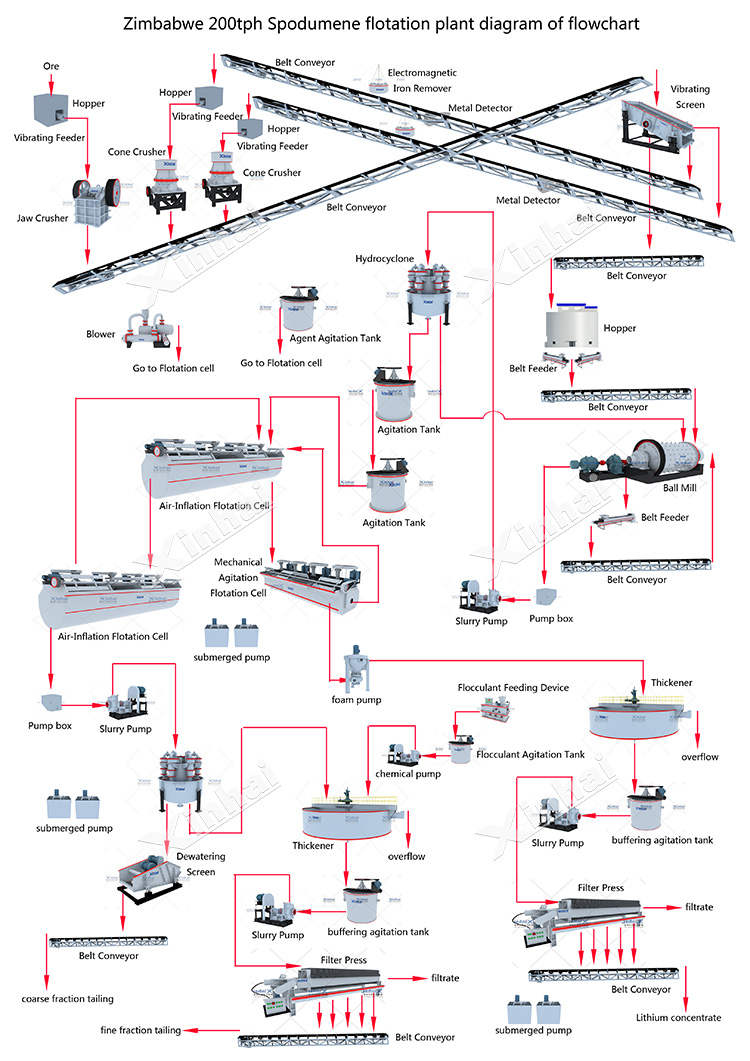
BackThe conventional process for extracting lithium from spodumene involves the following steps:
Mining and Crushing: Spodumene ore is mined from pegmatite deposits and then crushed to reduce its particle size.
Concentration: The crushed ore is subjected to gravity concentration, froth flotation, or a combination of both to separate spodumene from other minerals present in the ore.
Calcination: The spodumene concentrate is heated in a rotary kiln or fluidized bed calciner at temperatures between 1000-1100°C to convert the alpha-spodumene to beta-spodumene, a more reactive form that facilitates lithium extraction.
Leaching: The calcined spodumene is mixed with sulfuric acid (H2SO4) to form lithium sulfate (Li2SO4) and other byproducts. The slurry is then subjected to a solid-liquid separation process.
Purification: Impurities, such as aluminum and iron, are removed from the lithium sulfate solution using various techniques, including precipitation, solvent extraction, or ion exchange.
Precipitation: Lithium carbonate (Li2CO3) is precipitated from the purified lithium sulfate solution by adding sodium carbonate (Na2CO3) or another suitable reagent.
Drying and Packaging: The lithium carbonate precipitate is filtered, dried, and packaged for sale or further processing into lithium hydroxide, metal, or other lithium compounds.
Each stage's commonly used lithium extraction equipment has been discussed before.
03Innovations in Spodumene Lithium Extraction
BackSeveral innovations have been introduced to improve the efficiency, cost-effectiveness, and environmental footprint of the spodumene lithium extraction process. Some of these include:
Direct Lithium Extraction (DLE): This technology aims to extract lithium directly from spodumene ores without the need for calcination and leaching steps. DLE uses advanced techniques, such as selective ion-exchange, nano-filtration, or solvent extraction, to separate lithium from the ore effectively. This method reduces energy consumption, water use, and greenhouse gas emissions associated with the traditional process.
Alternative Leaching Methods: Researchers are exploring alternative leaching methods to replace sulfuric acid, which can be corrosive and harmful to the environment. These alternatives include hydrochloric acid, nitric acid, and organic acids, which may offer better selectivity and lower environmental impacts.
Process Optimization: Ongoing efforts are being made to optimize the entire spodumene lithium extraction process, including mining methods, ore processing techniques, and waste management practices. These efforts aim to minimize energy consumption, water use, and environmental impacts.
04Challenges in Spodumene Lithium Extraction
BackDespite the advancements in spodumene lithium extraction, several challenges remain:
Environmental Concerns: The traditional spodumene lithium extraction process generates significant amounts of waste and consumes large quantities of water and energy. Furthermore, the use of hazardous chemicals, such as sulfuric acid, can pose environmental and safety risks.
Economic Viability: The cost of lithium extraction from spodumene can be high, particularly in regions with limited infrastructure or high energy costs. This has led to the exploration of alternative lithium sources, such as brines, which can be more cost-effective in some cases.
Resource Availability: The availability of high-quality spodumene deposits may become a limiting factor as lithium demand continues to grow. This could necessitate the development of new extraction methods that can efficiently process lower-grade ores.
05Conclusion
BackThe spodumene lithium extraction process plays a critical role in meeting the growing global demand for lithium. Innovations in this field have the potential to improve efficiency, reduce costs, and minimize the environmental impacts associated withlithium production. However, several challenges remain, including environmental concerns, economic viability, and resource availability. Continued research and development efforts are essential to address these challenges and ensure the sustainable growth of the lithium industry. As innovations continue to emerge, the spodumene lithium extraction process will likely evolve, paving the way for a cleaner and more efficient lithium production landscape.
 +86 183 3575 8886
+86 183 3575 8886 pinklaurabao@gmail.com
pinklaurabao@gmail.com




 Message
Message Chat Now
Chat Now



















